|
Write the ionic equation by breaking all the soluble ionic compounds (those marked with an (aq)) into their respective ions. Most elements and covalent compounds (covalent compounds are formed when two or more nonmetallic elements are bonded to each other) are insoluble in water and should be shown with an (s), (l) or (g). Soluble ionics are identified with an (aq), insoluble ones with an (s). Use the solubility rules to determine which of the ionic compounds are soluble in water. Write the molecular equation and balance it.ĭetermine the state of each substance (gas, liquid, solid, aqueous). Each step will be demonstrated using the reaction of magnesium metal with hydrochloric acid as an example. To write ionic equations follow the steps below. While ionic equations show all of the substances present in solution, a net ionic equation shows only those that are changed during the course of the reaction. To calculate the membrane potential when two or more ions play a role, use the Goldman-Hodgkin-Katz (GHK) equation.Ionic equations and net ionic equations are usually written only for reactions that occur in solution and are an attempt to show how the ions present are reacting. 
Membrane permeability for an ion is defined as the relative ease with which the ion crosses the membrane, and is proportional to the total number of open channels for that ion in the membrane. The relative contribution of each ion is governed not only by its concentration gradient and valence, but also by its relative permeability. Instead, the value of V m will be determined by the relative contribution of each ion to the membrane potential. In this case, V m will not be at the equilibrium potential for either ion and, thus, no ion will be at equilibrium. If channels for two or more ions are present (and are open), both ions contribute to the membrane potential. Of course, it must be assumed that the ion channels are open in order to allow transmembrane movement of the ionic species for which the channel is specific. If only one ionic species is present in the system and/or only one type of ion channel is present, the Nernst potential also determines the resulting membrane potential ( V m). The Nernst equation can be derived based on simple thermodynamic principles (see lecture notes on the Resting Membrane Potential and Derivation of the Nernst Equation).Īs mentioned above, the Nernst equation calculates the equilibrium potential ( V Eq.) for an ion based on the charge on the ion (i.e., its valence) and its concentration gradient across the membrane.Typically, but not always, the concentrations are noted in mM. Note that the concentration unit must match that of out. in is the concentration of the ionic species X in the intracellular fluid.Note that the concentration unit must match that of in. out is the concentration of the ionic species X in the extracellular fluid.F is the Faraday's constant and is equal to 96485 C.mol -1 (Coulombs per mole).For example, z is +1 for Na +, +1 for K +, +2 for Ca 2+, -1 for Cl -, etc. z is the valence of the ionic species.T is the temperature in Kelvin (K = ☌ + 273.15).R is the universal gas constant and is equal to 8.314 J.K -1.mol -1 (Joules per Kelvin per mole). 
In this case, use the Goldman-Hodgkin-Katz (GHK) equation to calculate the V m. If two or more ions contribute to the membrane potential, the Nernst potential no longer yields the V m. 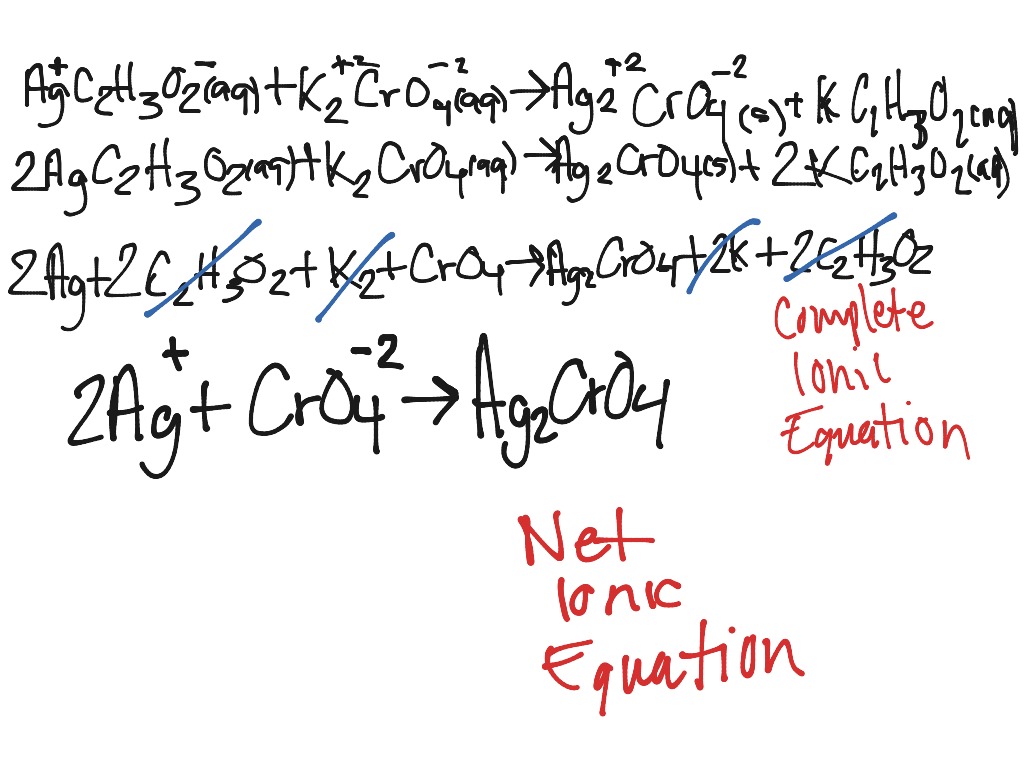
However, the equilibrium potential is typically reported in millivolts (mV). will also be the membrane potential ( V m). If only one ionic species is present in the system, and/or channels for only one ionic species are present (and open), then V Eq. It is common to use the ion symbol as a subscript to denote the equilibrium potential for that ion (e.g., V K, V Na, V Cl, V Ca, etc.). is the equilibrium potential (Nernst potential) for a given ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
